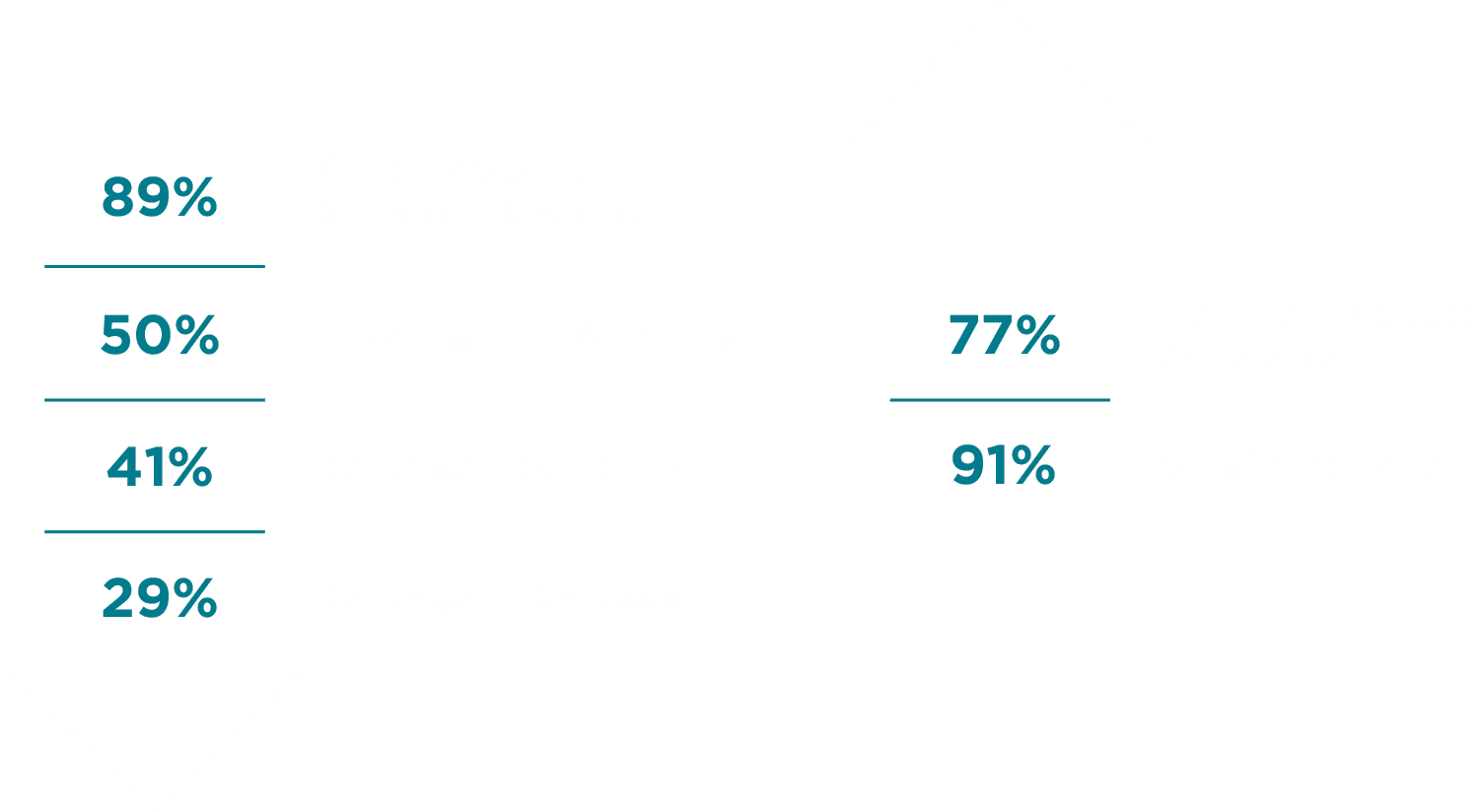Relief for Veterans
Freespira is a use-at-home medical device that is FDA-cleared to treat panic and PTSD symptoms. In peer-reviewed clinical studies, Freespira’s 28-day treatment has been shown to significantly reduce panic attacks and PTSD symptoms.
Patients complete two 17-minute sessions a day for 28 days. During each session, a sensor measures breathing patterns and provides real-time feedback. Patients who complete treatment will learn a new grounding and breathing technique to help manage symptoms associated with PTSD and panic disorder.
Please consult your VA clinician to determine if Freespira is right for you.
HOW FREESPIRA HELPS
Living with panic attacks or PTSD symptoms is hard
Patients who have completed Freespira’s 28-day treatment experienced symptom reduction:
The Results
Clinically demonstrated to help Veterans
A published study conducted at the VA Palo Alto Health Care System showed 89% of patients enrolled achieved clinically significant symptom reduction.3
Ostacher, et al., 2021
SURVEY RESULTS
Veterans report sleep problems, panic attacks and anxiety as most troubling symptoms4
The symptoms survey respondents say they most want to resolve:
0%
Inability to stay calm in difficult life situations
0%
Difficulty being present with family and friends
0%
Experiencing panic attacks and general anxiety
0%
Not sleeping consistently through the night
0%
Problems controlling anger or frustration
Freespira is easy to use, with 77% average adherence to the twice-a-day treatment over the 28-day protocol among participants in a study by VA researchers (Ostacher et al., 2021).
Freespira is shipped directly to the patient's home after it has been authorized by the VA.
Freespira is the only FDA-cleared, medication-free digital therapeutic treatment clinically demonstrated to reduce or eliminate PTSD symptoms and panic attacks.
"Since completing Freespira, I am able to control my anger, and I am not as depressed anymore. I've learned to cope better and am functioning well. I enjoy family activities again, and my family enjoys being around me, too. Freespira gave me an outlet to control my symptoms so I'm able to function in everyday life."
—Gary, Sergeant First Class
United States Army, Retired
get started
Relief for Veterans at the ready
Freespira can be used with talk therapy and medication.
After authorization, veterans will receive the Freespira system, which includes a sensor, a small breath-sampling tube (cannula), and a tablet with an app.
The treatment corrects irregular breathing patterns—underlying physiologic factors with PTSD and repeated anxiety attacks—and trains the body to reduce or eliminate them.
Freespira is listed on the Federal Supply Schedule 65IIA: V797D-50450, through our SDVOSB partner, Lovell Government Services.
Vendor Information:
CAGE: 71G35
DUNS: 079230337
UEI: MNH3STM1FTS6
Resources for Veterans
INFORMATION GUIDE
Benefit Overview
for Veterans
BLOG ARTICLE
3 Big Ideas about Veterans, PTSD, & Nontraditional Therapies